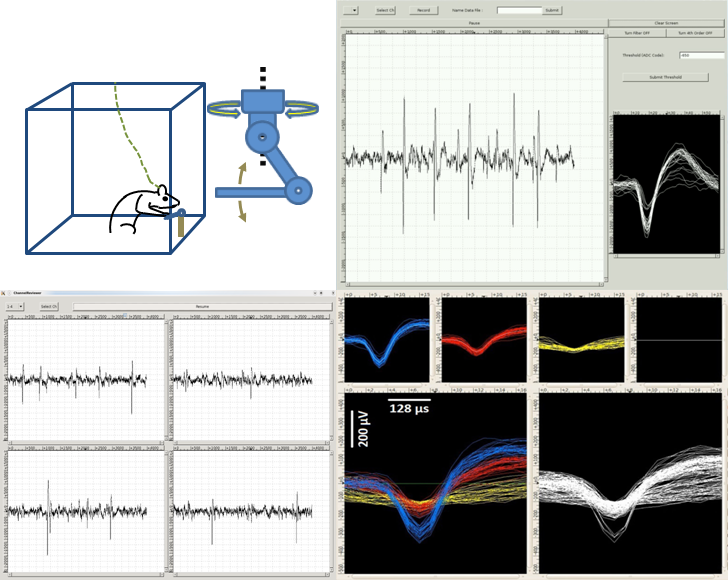
Neurophysiology of motor and neuroprosthetic skill learning
How are the motor related brain subsystems are involved in motor and neuroprosthetic skill learning? Motor skill describes the ability to move the muscles and limbs precisely in order to perform a voluntary action. Neuroprosthetic skill defines the ability to control the devices directly interacting with the brain tissue. It is known that brain's adaptation is crucial in both motor and neuroprosthetic skill learning. We develop new behavioral paradigms for rodents and study the adaptation mechanisms of the brain to the introduced neuroprosthetic devices. We utilize chronically implantable microelectrode arrays for recording the neural activity in the brain of behaving rodents.
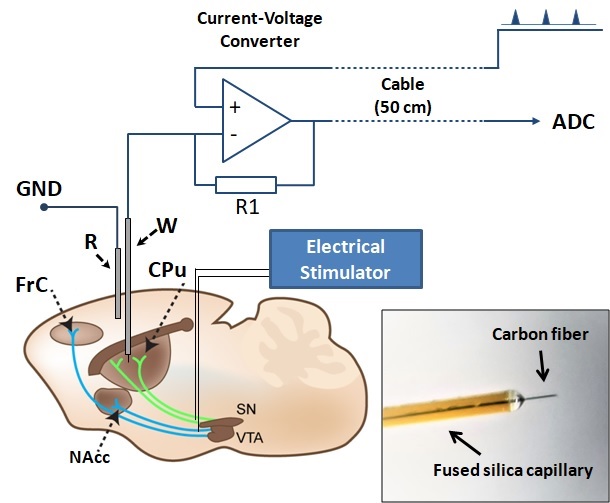
Physiology of the dopaminergic system
What is the role of dopaminergic and reward system in motor and neuroprosthetic skill learning? We utilize voltammetry, electrical stimulation and chronically implantable microelectrodes in order to measure dopamine transients in behaving rodents.
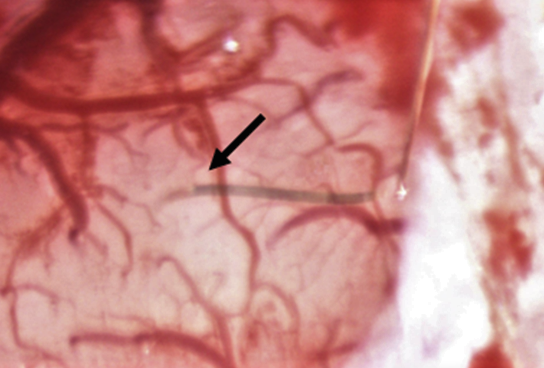
Neurotechnologies for clinical applications and research
How can we improve the neural interfaces for delivering seamless neuroprosthetic devices for the individuals suffering from neurological diseases and disorders? Recent studies indicate that microelectrode arrays made of ultraflexible materials can form reliable neural integration. Using this design approach based on utilization of ultraflexible materials we target to develop new neural interfaces which can enable long-term in vivo monitoring of both electrical activity of neurons and concentration of neurochemicals.
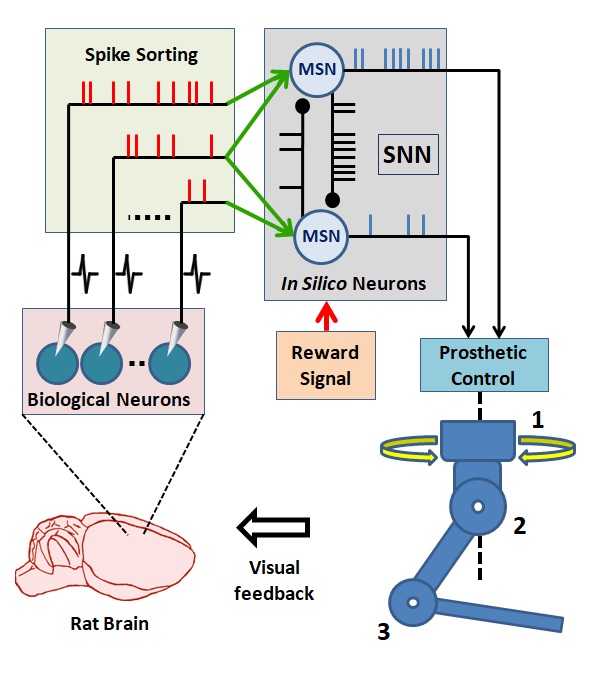
Bioinspired algorithms for neuroprosthetic control
How can we improve the neural decoders to provide high performance motor neuroprosthetic control? We believe that we can develop biologically inspired neuroprosthetic controllers. The biologically inspired neuroprosthetic controllers could be formed using a spiking neural network (SNN). In such a control paradigm, the SNN would consist of biologically plausible model neurons and receive simulated synaptic inputs from the extracellularly recorded cortical neurons as shown in the next figure. We believe that this SNN-based design approach has the potential to bring several advantages in neuroprosthetic system control, adaptation, and implementation. First, the information encoded by spike timing could be used at the input layer of the SNN-based neuroprosthetic controller. Second, spike timing plays a critical role in neuroplasticity, which is essential in neuroprosthetic learning. Therefore, the SNN-based neuroprosthetic controllers updating their parameters by simulating mechanisms of spike timing-dependent plasticity might have superior adaptation performance than existing firing rate-based neuroprosthetic systems. Third, implementation of the SNN-based neuroprosthetic controllers into neuromorphic chips can enable delivery of fully implantable, ultra low-power neuroprosthetic systems for paralyzed patients. For detailed information please refer to our publicaton titled: Toward building hybrid biological/in silico neural networks for motor neuroprosthetic control